.png)
The emission spectrum of atomic hydrogen is divided into a number of spectral series, with wavelengths given by the Rydberg formula: \frac.\): Dalton's symbols. It could not explain the varying intensity of emission lines. Even for hydrogen, a more accurate study of the observed spectra showed multiple components in some lines which could not be explained on the basis of this model. Hydrogen Bohr model was based on some assumptions which are listed below: The singular proton of the hydrogen atom or the protons and neutrons of the hydrogen-like species (ex- He +, Li 2+, etc. Bohr’s model for hydrogen atoms could not explain the line spectra of atoms other than hydrogen. Hydrogen Spectrum: Spectral lines of Hydrogen, divided into series. Bohr’s Atomic Model was for the hydrogen atom and hydrogen-like species (species with only one electron). Thus, Bohr worked on atomic spectra by assuming quantized electronic energy. As Rutherfords model fails to explain the hypothetical results regarding the atomic spectrum of elements. Bohr's model calculated the following energies for an electron in the shell, n : E ( n) 1 n 2 13. Postulates of Bohrs atomic model: Bohr assumed that electromagnetic radiation has dual nature which means that radiations have both wave/particle-like properties. Similarly, the emission spectra of molecules can be used in chemical analysis of substances. Key points Bohr's model of hydrogen is based on the nonclassical assumption that electrons travel in specific shells, or orbits, around the nucleus. All substances, according to Dalton’s atomic theory, are made up of atoms, which are indivisible and. It stated that all matter was made up of small, indivisible particles known as ‘atoms’. Each element’s emission spectrum is unique, and therefore spectroscopy can be used to identify elements present in matter of unknown composition. Dalton’s atomic theory was a scientific theory on the nature of matter put forward by the English physicist and chemist John Dalton in the year 1808. 
As a theory, it can be derived as a first-order approximation of the hydrogen atom using the broader and much more accurate quantum mechanics and thus may be considered to be an obsolete scientific theory. The emission spectrum of a chemical element or chemical compound is the spectrum of frequencies of electromagnetic radiation emitted by an atom’s electrons when they are returned to a lower energy state. The Bohr model is a relatively primitive model of the hydrogen atom, compared to the valence shell model. emission: In a spectral sense, what occurs when an electron transitions between a higher energy level and a lower one, resulting in the release of a photon of predictable energy.spectrum: A range of colors representing light (electromagnetic radiation) of contiguous frequencies hence electromagnetic spectrum, visible spectrum, ultraviolet spectrum, etc.Further series are unnamed but follow exactly the same pattern as dictated by the Rydberg equation. Niels Bohr (born October 7, 1885, Copenhagen, Denmarkdied November 18, 1962, Copenhagen) Danish physicist who is generally regarded as one of the foremost physicists of the 20th century. 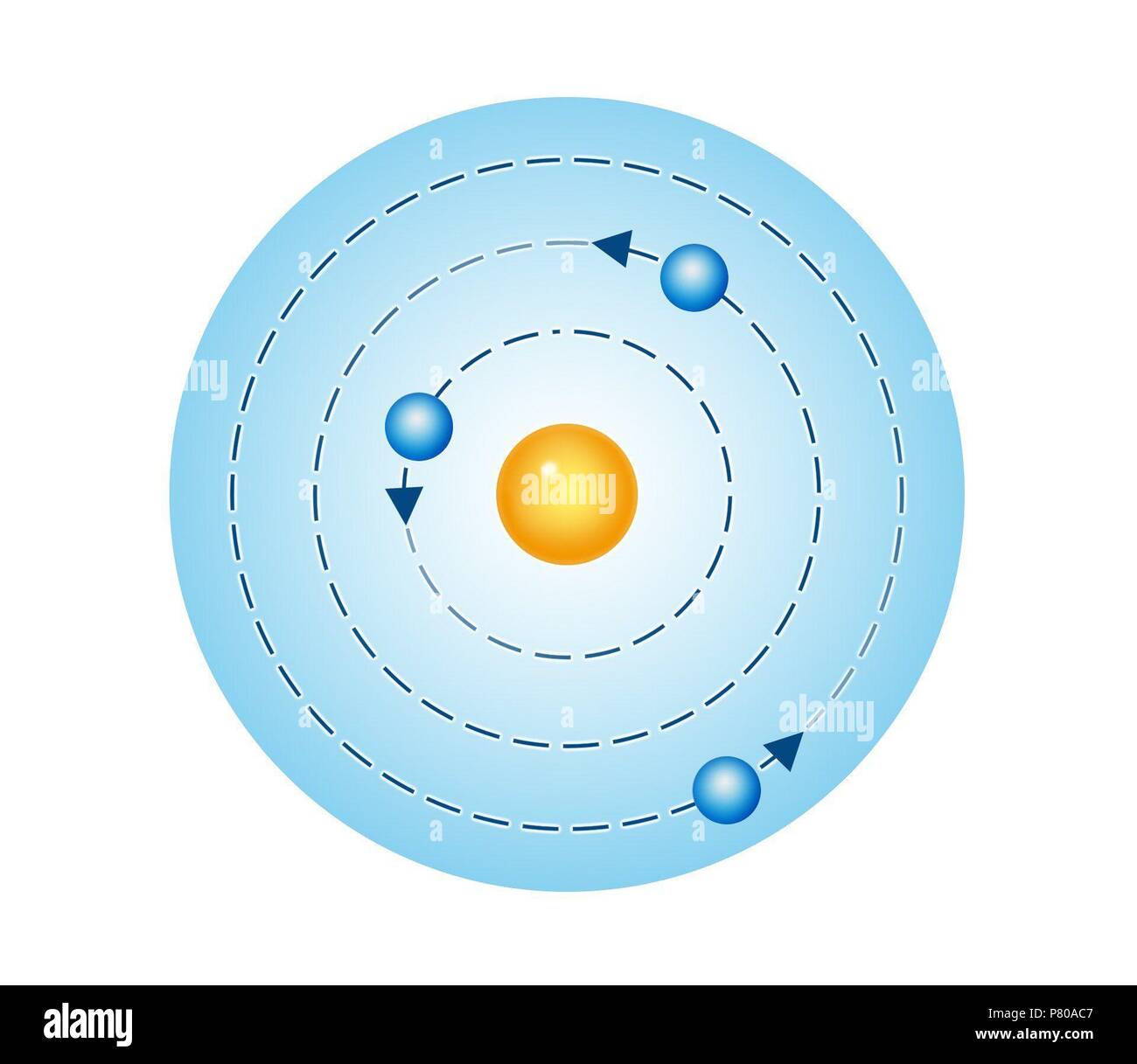
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
